RESTORATION ECOLOGY
Assessing the fitness of the hatchery Atlantic salmon (Salmo salar) population and the genetic composition of the extant naturally spawning population as a support to the development of stock restoration strategies in the river Shannon
Some of the most productive Atlantic salmon rivers in Europe have been harnessed for hydro-electric power generation e.g. Shannon (Ireland), Conan (Scotland), Linares (Spain). In Ireland alone some 35% of the potential salmon producing habitat is impounded above hydroelectric dams. In compensation, hatchery mitigation programmes were established in most of these rivers, in order to compensate for the loss of productivity, to maintain natural runs and to preserve biodiversity. Despite the best efforts of these hatchery programmes, many of the salmon populations above these facilities are effectively extinct. The large hatchery programmes continue to exist but are increasingly coming under the spotlight from cost benefit analyses and their success in maintaining fisheries and protecting biodiversity.
Most of these mitigation schemes were developed many decades ago before much of the contemporary information about sub-specific population genetics was developed. Thus, it would seem timely to reassess and redirect mitigation programmes with respect to the large body of evolutionary, population and quantitative genetic knowledge that now exists, particularly in terms of metapopulation theory, landscape genetics and new knowledge about the biology of the salmon (effective population size etc.). It might also be possible to simulate natural re-colonisation processes, by combining ecological and evolutionary biological principles to resolve these most difficult fisheries management problems.
There are two parts to this project (1) Common Garden Experiment and (2) Establishment of a Genetic Baseline
Common Garden Experiment
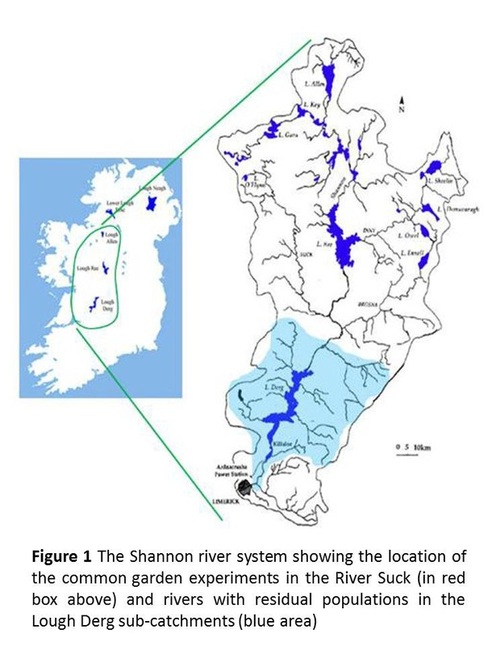
The first part is an assessment of the relative fitness of the progeny of the Shannon hatchery population with respect to the progeny of the contemporary extant naturally-spawning population and the progeny of two non-impacted wild salmon populations obtained from rivers located below the dam. We have completed the establishment of an experimental population of approximately 378000 eggs from 125 families. An equal proportion of these eggs was introduced into two parallel headwater streams on the River Suck, a major tributary of the River Shannon (March 2011). We sampled approximately 2,000 of the surviving fry at the end of the summer 2011. We will collect a further 500 parr surviving at the end of the second summer (2012). We have undertaken an assessment of the performance of individual families using genetic parentage assignment. Parr collected for genetic sampling in 2012 will be fitted for PIT tags at the same time as they are sampled for genetics, and PIT tag reading stations are being established on one of the experimental rivers to enable estimates of family and group survival to the sea-going smolt stage. In addition three rotary screw traps (Fig. 3) have been deployed in the Bunowen to facilitate the capture and tissue sampling of one year old smolts migrating in 2012 and to enable mark recapture estimates of population size.
Some of the most productive Atlantic salmon rivers in Europe have been harnessed for hydro-electric power generation e.g. Shannon (Ireland), Conan (Scotland), Linares (Spain). In Ireland alone some 35% of the potential salmon producing habitat is impounded above hydroelectric dams. In compensation, hatchery mitigation programmes were established in most of these rivers, in order to compensate for the loss of productivity, to maintain natural runs and to preserve biodiversity. Despite the best efforts of these hatchery programmes, many of the salmon populations above these facilities are effectively extinct. The large hatchery programmes continue to exist but are increasingly coming under the spotlight from cost benefit analyses and their success in maintaining fisheries and protecting biodiversity.
Most of these mitigation schemes were developed many decades ago before much of the contemporary information about sub-specific population genetics was developed. Thus, it would seem timely to reassess and redirect mitigation programmes with respect to the large body of evolutionary, population and quantitative genetic knowledge that now exists, particularly in terms of metapopulation theory, landscape genetics and new knowledge about the biology of the salmon (effective population size etc.). It might also be possible to simulate natural re-colonisation processes, by combining ecological and evolutionary biological principles to resolve these most difficult fisheries management problems.
There are two parts to this project (1) Common Garden Experiment and (2) Establishment of a Genetic Baseline
Common Garden Experiment
The first part is an assessment of the relative fitness of the progeny of the Shannon hatchery population with respect to the progeny of the contemporary extant naturally-spawning population and the progeny of two non-impacted wild salmon populations obtained from rivers located below the dam. We have completed the establishment of an experimental population of approximately 378000 eggs from 125 families. An equal proportion of these eggs was introduced into two parallel headwater streams on the River Suck, a major tributary of the River Shannon (March 2011). We sampled approximately 2,000 of the surviving fry at the end of the summer 2011. We will collect a further 500 parr surviving at the end of the second summer (2012). We have undertaken an assessment of the performance of individual families using genetic parentage assignment. Parr collected for genetic sampling in 2012 will be fitted for PIT tags at the same time as they are sampled for genetics, and PIT tag reading stations are being established on one of the experimental rivers to enable estimates of family and group survival to the sea-going smolt stage. In addition three rotary screw traps (Fig. 3) have been deployed in the Bunowen to facilitate the capture and tissue sampling of one year old smolts migrating in 2012 and to enable mark recapture estimates of population size.

Preliminary results from the parentage assignment of samples collected from the 0+ sample collected in 2011 show the local Shannon populations (both hatchery and feral) surviving best in comparison to the progeny of the broodstock sourced from rivers below the dam and outside the system. This will have important implications for the future management of the salmon re-establishment programme in the Shannon.
Establishment of a Genetic Baseline
The second part is the provision of a genetic baseline consisting of: samples collected from (1) historical populations derived from archive scale collections (1929–1980); (2) contemporary residual naturally- spawning populations from above the hydroelectric generating facility; (3) the contemporary hatchery population; and (4) contemporary samples from wild populations in rivers located below the dam. In 2010, we collected material from five residual populations in tributary rivers flowing into Lough Derg and the two principal rivers downstream of the hydroelectric facility. In addition, we have been undertaking some trial screening of archive scales samples to assess DNA quality and to determine the most effective method of acquiring high quality DNA.
Establishment of a Genetic Baseline
The second part is the provision of a genetic baseline consisting of: samples collected from (1) historical populations derived from archive scale collections (1929–1980); (2) contemporary residual naturally- spawning populations from above the hydroelectric generating facility; (3) the contemporary hatchery population; and (4) contemporary samples from wild populations in rivers located below the dam. In 2010, we collected material from five residual populations in tributary rivers flowing into Lough Derg and the two principal rivers downstream of the hydroelectric facility. In addition, we have been undertaking some trial screening of archive scales samples to assess DNA quality and to determine the most effective method of acquiring high quality DNA.
